Study demonstrates clinical value of the Cunningham Panel™ and accuracy in patients with symptoms of an autoimmune encephalopathy
Antineuronal antibodies correlate with pre- and post-treatment neuropsychiatric symptoms
This newly published case series examined 58 patients tested with the Cunningham Panel™ who were diagnosed with the infection-triggered autoimmune encephalopathy — Pediatric Autoimmune Neuropsychiatric Disorder Associated with Streptococcal infections (PANDAS) or Pediatric Acute-onset Neuropsychiatric Syndrome (PANS).
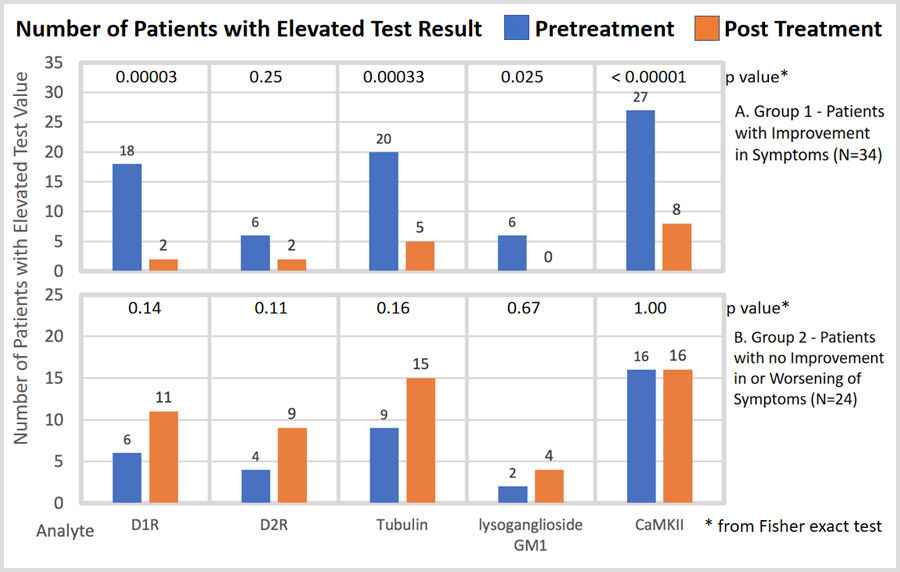
The authors examined whether changes in pre- and post-treatment symptoms correlated with anti-neuronal antibody titers and the neuronal cell stimulation assay measured by the Cunningham Panel™.
“This study revealed a strong positive association between changes in neuropsychiatric symptoms and changes in the level of antineuronal antibodies and antibody-mediated CaMKII human neuronal cell activation,” said Shimasaki.
Additionally, this study evaluated the Panel’s clinical performance for reproducibility and reliability. The authors also examined whether these assays might serve as biological evidence for monitoring treatment response in this patient population and assist in predicting effectiveness of antimicrobial and immunomodulatory therapy.
“These study findings support clinical utility of the antineuronal and cell-stimulatory assays comprising the Cunningham Panel™ as an aid in diagnosis, and laboratory evidence supporting an underlying autoimmune etiology,” 1 the authors concluded.
Highlights:
- The Cunningham Panel™ assays, which measure antineuronal antibodies against Dopamine D1 Receptor, Dopamine D2L Receptor, Lysoganglioside-GM1, Tubulin, and a cell-stimulation CaMKII assay, demonstrated test-retest reproducibility. Repetitive testing of multiple samples, over a period of time, demonstrated assay robustness and reproducibility.
- In comparing pre- and post-treatment patient symptom status, it was revealed that the Cunningham Panel™ results paralleled changes in a patient’s neuropsychiatric symptoms following treatment. The Cunningham Panel demonstrated an overall accuracy of 86% to 90%, a sensitivity of 88% and a specificity of 83% to 92% when comparing clinical symptoms to test results.
- The Cunningham Panel™ test results can provide clinicians with biological evidence supporting their diagnosis of an immune-mediated disorder, which may support the modification of a treatment modality.
- The study results support the clinical utility of the Cunningham Panel™ as an aid to a physician’s diagnosis and may help in managing treatment of an infection-triggered autoimmune encephalopathy, such as PANS/PANDAS.
This video presentation reviews study evaluating accuracy and sensitivity of the Cunningham Panel for an infection-triggered autoimmune encephalopathy.
Data highlights value and reliability of Cunningham Panel in aiding diagnosis and treatment of autoimmune encephalopathy, characterized by neuropsychiatric symptoms.
Physician’s Digital Information Packet
To learn more about the Cunningham Panel™ and its utility in assisting with the diagnosis of an autoimmune encephalopathy characterized by neuropsychiatric symptoms, such as PANS/PANDAS, we have compiled a list of journal articles and video clips that may be helpful.
Schedule Your Personal Phone Consultation
Learn more about how the Cunningham Panel™ can assist you in your diagnosis of treatable autoimmune encephalopathies characterized by abnormal neuropsychiatric behaviors. To schedule a personal phone consultation with a Moleculera Labs clinical staff member, please complete the form below.
- Craig Shimasaki, Richard E. Frye, Rosario Trifiletti, Michael Cooperstock, Gary Kaplan, Isaac Melamed, Rosalie Greenberg, Amiram Katz, Eric Fier, David Kem, David Traver, Tania Dempsey, M. Elizabeth Latimer, Amy Cross, Joshua P. Dunn, Rebecca Bentley, Kathy Alvarez, Sean Reim, James Appleman. Evaluation of the Cunningham Panel™ in pediatric autoimmune neuropsychiatric disorder associated with streptococcal infection (PANDAS) and pediatric acute-onset neuropsychiatric syndrome (PANS): Changes in antineuronal antibody titers parallel changes in patient symptoms. Journal of Neuroimmunology, In Press-15 February 2020.Online ahead of print https://www.jni-journal.com/article/S0165-5728(19)30352-2/fulltext