Several published studies demonstrate the sensitivity (rate of true positives), specificity (rate of true negatives), and accuracy of the Cunningham Panel™.
In a recently published clinical study, patients with neurologic, psychiatric and behavioral symptoms consistent with a PANS or PANDAS diagnosis, revealed that the Cunningham Panel™ results closely correlated with changes in a patient’s neuropsychiatric symptoms before and after treatment.
The Cunningham Panel™ demonstrated an overall accuracy of 86%, a sensitivity of 88%, and a specificity of 83% when comparing clinical symptoms to test results. 1
“These study findings support clinical utility of the antineuronal and cell-stimulatory assays comprising the Cunningham Panel™ as an aid in diagnosis, and laboratory evidence supporting an underlying autoimmune etiology,” the authors state.
Determining response to IVIG treatment
Connery et al. found that the Cunningham Panel™ predicted a patient’s positive response to intravenous immunoglobulin (IVIG) treatment with a sensitivity of 90% to 100%, a specificity of 67% to 75%, and an overall accuracy between 81% and 88%. 2
“Our study verifies previous reports that suggest that IVIG may be useful in individuals with ASD [autism spectrum disorder] who demonstrate biomarkers of immune abnormalities,” the authors state.
The Cunningham Panel™ was found to be effective in predicting treatment outcomes for a subset of patients with autism who also suffered from autoimmune encephalopathy.
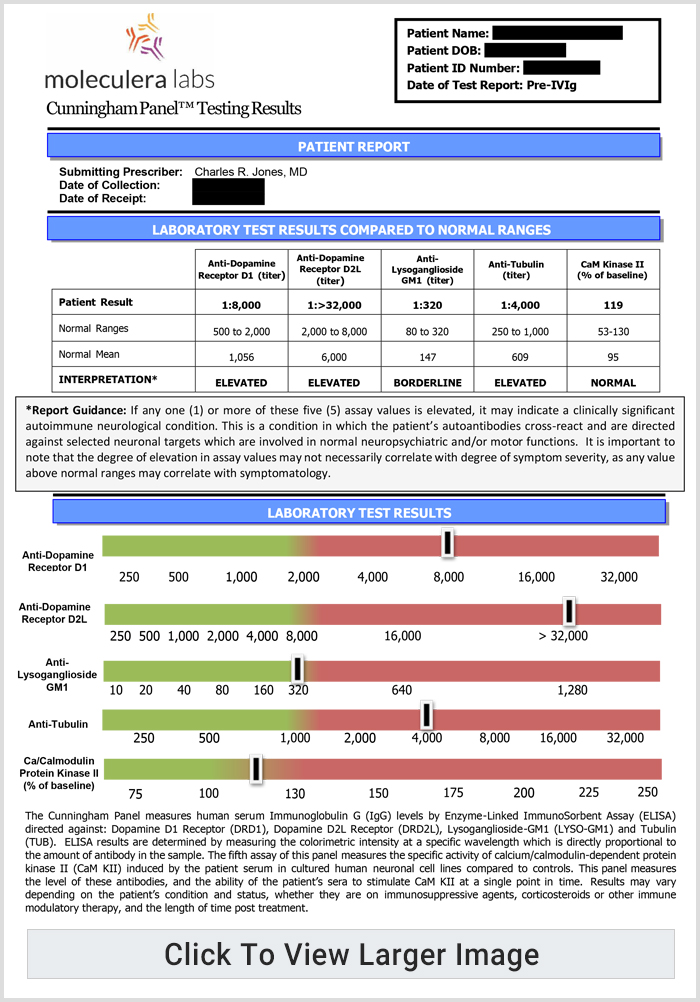
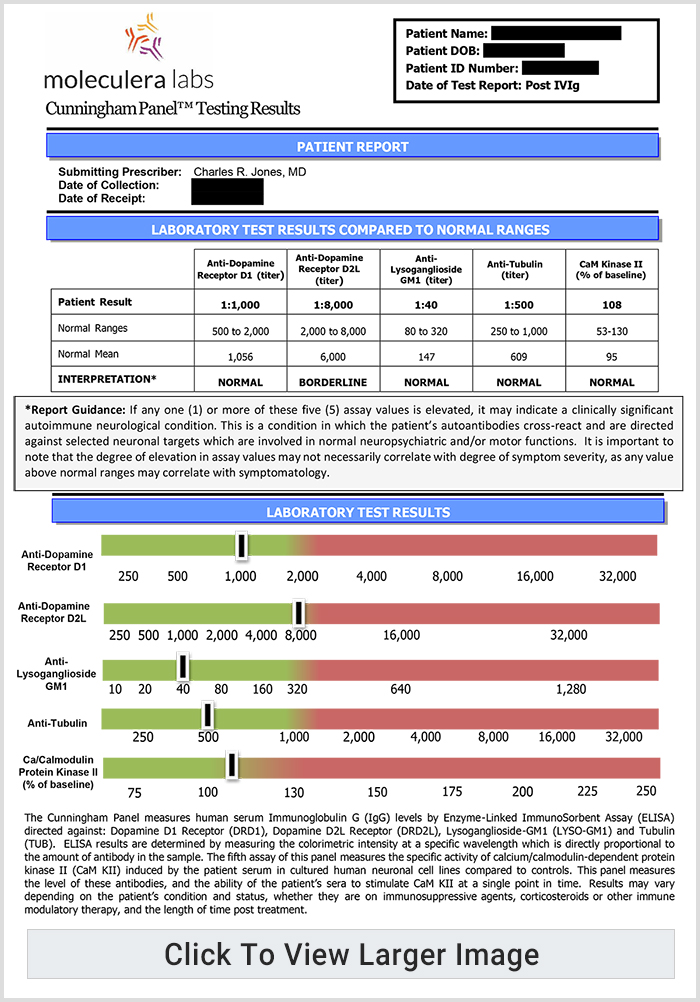
A Cunningham Panel showed elevations in anti-tubulin antibodies (2000; nl = 250–1000) and CaMKII (151, nl < 130), prompting a trial of IVIG at 1 g/kg/day × 2 days every month. Within 2 days of the IVIG treatment, his handwriting significantly improved. (See Fig. 6b).
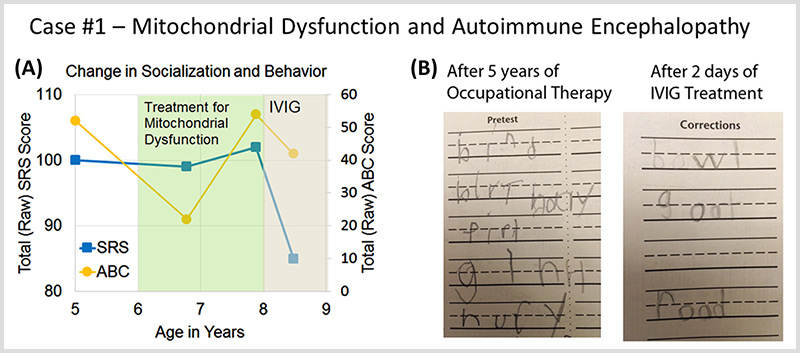
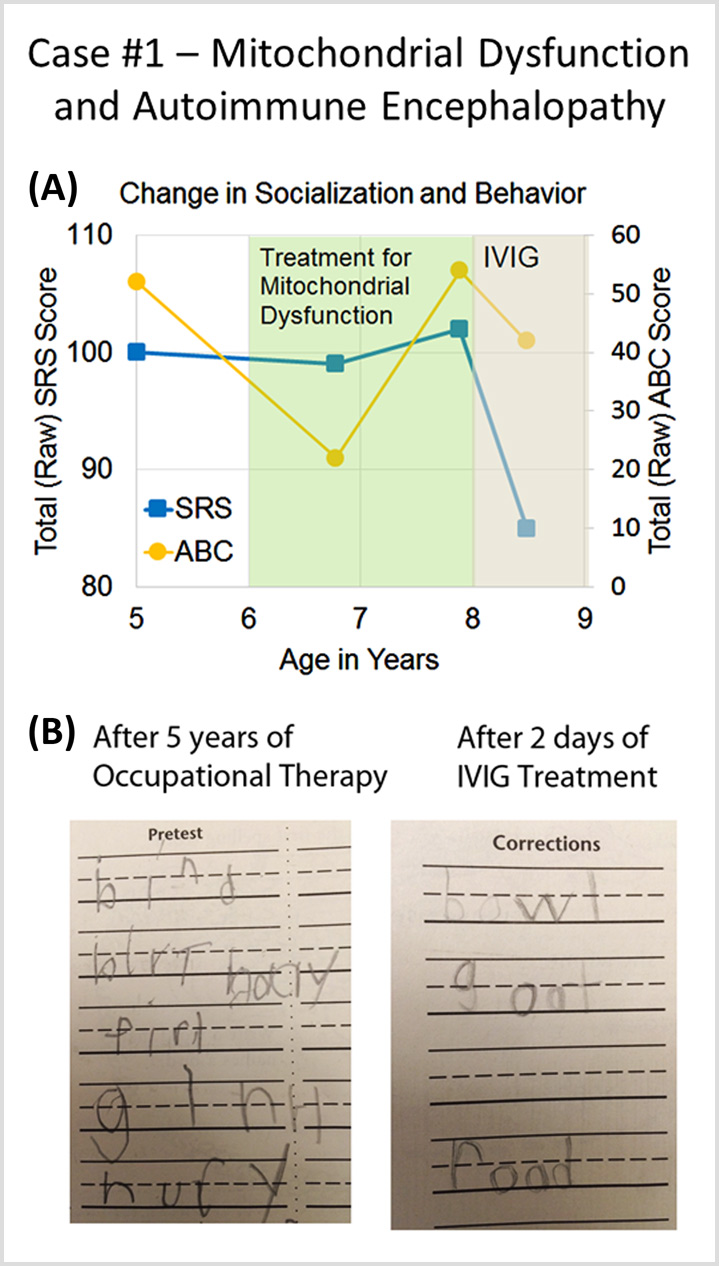
(Source: Connery, K., Tippett, M., Delhey, L.M. et al. Intravenous immunoglobulin for the treatment of autoimmune encephalopathy in children with autism. Transl Psychiatry 8, 148 (2018). https://doi.org/10.1038/s41398-018-0214-7)
Test identifies autoimmune encephalitis in children
Another study indicates antineuronal antibody biomarkers and CaMKII activation, comprising the Cunningham Panel™, may be a useful adjunct to clinical diagnosis of Sydenham chorea, PANDAS and related disorders and are the first known group of autoantibodies detecting dopamine receptor-mediated encephalitis in children. 4
Test Background
The Cunningham Panel™ is comprised of five high complexity metabolic tests. Four of these tests measure circulating levels of specific autoantibodies in a patient’s serum at the time the specimen is collected. Test results are expressed as a titer, or final dilution, at which an endpoint reaction was observed on an Enzyme-Linked Immunosorbent Assay (ELISA) format.
The fifth test, the Calcium-dependent Calmodulin Protein Kinase II (CaMKII) is a brain cell stimulation assay. The CaMKII test measures the ability of a patient’s autoantibodies to stimulate the CaMKII enzyme, leading to an upregulation of brain neurotransmitters, such as dopamine, epinephrine, and norepinephrine, which can disrupt neuronal functioning resulting in a variety of neurologic and/or psychiatric symptoms.
The Cunningham Panel™ was originally developed based on more than two decades of research conducted by Dr. Madeleine Cunningham at the University of Oklahoma, in conjunction with scientists from the National Institutes of Mental Health (NIMH).
Moleculera Labs is an accredited CLIA (Clinical Laboratory Improvement Amendment) and COLA (Commission on Laboratory Accreditation) laboratory (CLIA: 37D2082408; COLA: 25744) and is authorized to receive physician orders from all 50 states in the U.S. It is also available through our international partners.
WATCH VIDEO
- Shimasaki C, Frye RE, Trifiletti R, Cooperstock M, Kaplan G, Melamed I, Greenberg R, Katz A, Fier E, Kem D, Traver D, Dempsey T, Latimer ME, Cross A, Dunn JP, Bentley R, Alvarez K, Reim S, Appleman J. Evaluation of the Cunningham Panel™ in pediatric autoimmune neuropsychiatric disorder associated with streptococcal infection (PANDAS) and pediatric acute-onset neuropsychiatric syndrome (PANS): Changes in antineuronal antibody titers parallel changes in patient symptoms. J Neuroimmunol. 2020 Feb 15;339:577138. doi: 10.1016/j.jneuroim.2019.577138. Epub 2019 Dec 15. PMID: 31884258.
- Connery, K. et al. Intravenous immunoglobulin for the treatment of autoimmune encephalopathy in children with autism. Transl. Psychiatry 8, 148 (2018). doi: https://pubmed.ncbi.nlm.nih.gov/30097568/
- Chain Jennifer L., Alvarez Kathy, Mascaro-Blanco Adita, Reim Sean, Bentley Rebecca, Hommer Rebecca, Grant Paul, Leckman James F., Kawikova Ivana, Williams Kyle, Stoner Julie A., Swedo Susan E., Cunningham Madeleine W. “Autoantibody Biomarkers for Basal Ganglia Encephalitis in Sydenham Chorea and Pediatric Autoimmune Neuropsychiatric Disorder Associated With Streptococcal Infections.” Frontiers In Psychiatry, vol. 11, 2020, p. 564., doi: https://www.frontiersin.org/article/10.3389/fpsyt.2020.00564
Learn More About The Cunningham Panel™
Overview Of The Cunningham Panel™
What does the Cunningham Panel™ measure?